滴雞精的健康旅程,從實驗到體驗的一步一腳印
最一開始接觸滴雞精,說不上是誰先起頭,大概就是某個週末聊天時Milo帶來的吧,他總是會突然冒出新東西。那天我們三個人窩在小客廳,桌上放著幾包不知名牌子的雞湯萃取液,也有幾罐標示看起來很專業的品牌。Zin好像對這類產品一直半信半疑,她覺得廣告裡演得太誇張。我倒是沒想那麼多,只覺得身體偶爾累、免疫力這種東西也許真該試著補一下。剛開始喝,其實沒什麼感覺,有時候甚至分不清到底是心理作用還是真的有差。記錄過程中偶爾會忘了哪一天喝過、哪一天只是打開聞味道,感覺生活和健康之間,好像比我以為的還要黏在一起。
簡化選擇過程,如何用三步驟找到理想產品
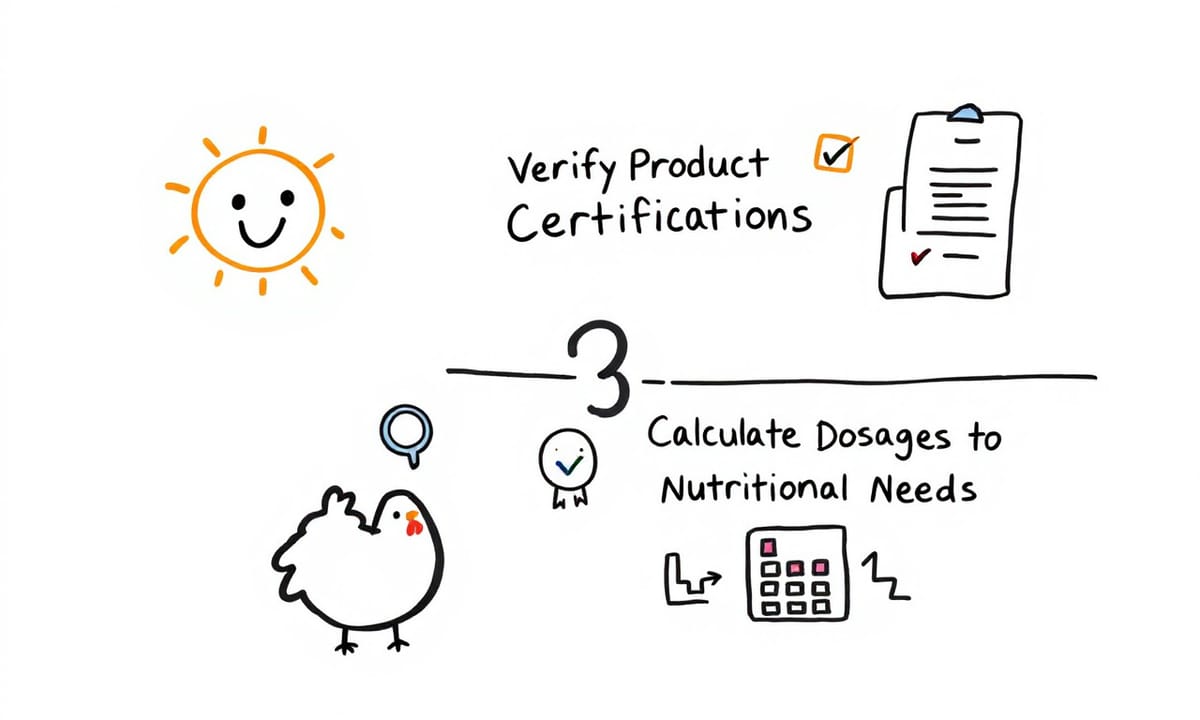
挑選滴雞精猶如解開一道難題,Milo巧妙地發展出一套簡單又有效的篩選方法。他認為,只要掌握三個關鍵步驟,就能輕鬆找到最適合自己的產品。
首先是確認個人過敏原,避免誤踩雷區。接著要查驗產品認證,確保來源可靠。最後則是仔細換算劑量,對應個人的營養需求。這套方法猶如一張篩選地圖,讓原本複雜的選購過程變得清晰明確。
不再為眾多品牌猶豫不決,也不必擔心買到不合適的產品。透過這三步驟,消費者彷彿擁有一把精準的選購金鑰,輕鬆穿越產品叢林,直達理想目標。
首先是確認個人過敏原,避免誤踩雷區。接著要查驗產品認證,確保來源可靠。最後則是仔細換算劑量,對應個人的營養需求。這套方法猶如一張篩選地圖,讓原本複雜的選購過程變得清晰明確。
不再為眾多品牌猶豫不決,也不必擔心買到不合適的產品。透過這三步驟,消費者彷彿擁有一把精準的選購金鑰,輕鬆穿越產品叢林,直達理想目標。
Comparison Table:
| 檢核項目 | 詳細說明 |
|---|---|
| 過敏原確認 | 仔細查看產品包裝上的過敏原成分,避免引發個人過敏反應。 |
| 官方認證查驗 | 確認產品的正式認證標籤,以確保來源可靠性和品質。 |
| 劑量換算 | 根據個人需求進行劑量換算,選擇最適合自己的產品規格。 |
| 品牌與成分比較 | 評估不同品牌的蛋白質含量和配方簡單性,選擇符合自己健康需求的產品。 |
| 飲用方式調整 | 嘗試不同飲用時機和搭配,如空腹或飯後,以及添加其他配料以改善口感。 |
當心冒牌貨!學會辨識真正的滴雞精
這些年買過的滴雞精,回想起來,好像總有幾次不是踩到標示亂七八糟,就是挑了那些看起來很厲害卻喝不出效果的。Zin那次以為撿到便宜,結果買了一批味道怪怪的風味液,後來才發現根本不是正統滴製品;聽說別人也常遇到類似情況,有人一開始看包裝漂亮就下手,最後只能怪自己沒認清來源。有時候光靠廣告或者外觀真的判斷不了什麼,大概將近一半朋友都說早期曾經買錯過。細節上,有些廠牌名字取得很像、包裝顏色也差不多,一個轉念之間就容易誤會。等意識到其實喝進去的不一定真有營養,不如當初不要花那筆錢。
早晨儀式感:空腹飲用滴雞精的神奇效果
清晨陽光透過廚房窗簾灑落,Milo正專注地從冰箱取出一小瓶滴雞精。他習慣在早餐前這個時刻,靜靜地品嚐這份養生儀式。冰箱門輕輕關上,發出細微的聲響,彷彿預示著一天即將開始。他喜歡在胃還空虛、身體尚未完全甦醒的剎那,給自己一個溫和的營養補給。這個看似簡單的早晨小習慣,已經悄然成為他生活節奏中不可或缺的一部分,彷彿是喚醒身體機能的一種儀式。
包裝材質對營養的重要性,你知道嗎?
說到營養價值,大家總以為成分表寫得明白清楚就沒問題,但其實那些容易忽略的小細節才是左右效果的關鍵。像Milo選東西的方法,看似簡單到只有三步,其實每個細節都藏著眉角。他有次翻來覆去查了好幾份產品資訊,結果還是發現自己曾經沒注意過一個包裝上極小的標示。有些人總覺得買健康食品就是圖個安心,可明明外觀差不多,裡頭到底是不是真的適合自己?過敏原、產地來源這種資訊,有時候被塞在不起眼的地方,一不小心就漏看了。有人說認證標章很重要,也不是每回都能保證萬無一失,畢竟市面品牌數量太多,有的甚至連名字都會讓人搞混。劑量換算那件事,看起來像學校考試題目,大部分人根本懶得理。但偏偏這些累積下來的小判斷,很可能就改變了你喝下肚之後真正感受到什麼。不只是雞精啦,其它補給品也是差不多道理,誰會記得每次都細看呢?
為什麼滴雞精的胺基酸濃度如此關鍵?

解決腥味困擾,各種創意喝法讓口感升級!
「味道好腥怎麼辦?」這題在群組爆紅,討論串一滑有將近一半的人都說自己喝過滴雞精有點怕那股味兒。Milo之前在網路上看到,有人建議先聞一下再喝,但他覺得聞了反而更難下嚥。有人分享說低溫脫腥技術可以壓掉不少怪味,雖然不知道是不是每個牌子都有用到這種做法,也有人提到瓶身會貼個什麼脫氣標籤,好像是幫助保持新鮮?Zin反而沒那麼排斥,她嘗試加點檸檬草一起煮,說起來多了點香氣就不太在意原本的腥感。啊對,有人留言問哪種方式最有效,但其實答案很分歧,有些人只要換個時機、空腹還是飯後喝,體感就差很多,不見得全靠技術或配料——所以到底怎樣最好,好像也沒一定規則,只能多試幾種方法才知道適不適合自己。
把滴雞精當作日常充電器,你準備好了嗎?
選購滴雞精就像挑選手機配件,Milo發展出一套精簡的篩選邏輯。他把複雜的選擇過程,巧妙地濃縮成三個關鍵步驟:先看過敏原標示,就像檢查手機是否相容;再查驗官方認證,如同確認正版配件;最後換算劑量,彷彿調整充電器的輸出功率。這套方法讓原本瑣碎的選購,瞬間變得輕鬆直觀,猶如找到一款完美的智慧配件。
消費者該如何看清蛋白質含量差異?
琳瑯滿目的滴雞精,怎麼選才不會踩雷?有朋友說大品牌就一定可靠,但也有人跟我們聊過,包裝再精美的,有時候裡面根本不是純滴雞精。配方、濃度、來源地,這些資訊標示看似透明,可是到底哪一項最關鍵,好像也沒共識。有些人主張成分越單純越好,也有人偏愛添加各種草本,講得頭頭是道。網路上評價多到讓人眼花撩亂,同一款產品有人喝了說精神變好,有的卻覺得差不多。甚至有媒體報導提過,市售雞精蛋白質含量落差能到數十倍,那是不是蛋白質多就代表比較好?還是要看游離胺基酸?總之,每次站在貨架前,都忍不住想問:這麼多選擇,到底誰才真正能「滴」出健康?
懶人包全流程揭秘,簡單有效的保存與加熱方法
選購滴雞精看似簡單,其實大有學問。Milo總結出一套實用檢核表,只需三個步驟就能輕鬆篩選合適產品。首先,仔細確認包裝上的過敏原成分,避免誤踩雷區;接著查驗官方認證,確保產品來源可靠;最後則是精準換算劑量,根據個人需求選擇最適合的規格。這套方法不僅省去猜測的煩惱,更讓選購過程變得輕鬆直接,猶如解開一道簡單又實用的生活難題。




































